You are here
Species
Plasmodium
IUCN
NCBI
EOL Text
Organisms of the genus Plasmodium are defined as distinct from other Apicomplexa, and other organisms sometimes considered malaria parasites (Peréz-Tris et al. 2005), as parasites that both undergo merogony (multiple divisions of the nucleus followed by segmentation of the cytoplasm producing daughter cells called merozoites) in erythrocytes, and that produce hemozoin pigment, the crystalline by-product of hemoglobin digestion. Other members of the order Haemosporida vary in the combination of these characters. The family Haemoproteidae produces pigment, but merogony occurs in tissues other than erythrocytes. The families Garniidae and Leucocytozoidae do not produce pigment at any stages, but in the case of the former do show merogony in blood cells.

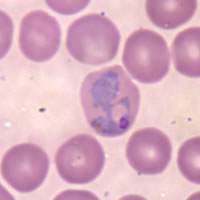
P. falciparum schizont stage parasite (center) showing merogony, recognizable by approximately 24 new merozoites identified by the Giemsa-stained nuclei (purple), and showing hemozoin pigment (yellow)
All malaria parasites have a sexual life stage that occurs in a blood-feeding insect, which is the definitive host for these organisms (also known as the “vector,” in epidemiology).
Traditionally, Plasmodium species were described based on morphological and morphometric characteristics, primarily of the blood stage of the lifecycle. Other characters used in the past for classification have been virulence-level and life-cycle period in the erythrocytic stage (24, 48, or 72 hours). Morphological characterization has often proven to be insufficient to distinguish species, and has also been shown to conflict with molecular sequence data. Scientists are beginning to use advanced methods and molecular characters for parasite taxonomy and phylogenetics. (See Discussion of Phylogenetic Relationships.)
| License | http://creativecommons.org/licenses/by-nc-sa/3.0/ |
| Rights holder/Author | Martine Zilversmit, Susan Perkins, Tree of Life web project |
| Source | http://tolweb.org/Plasmodium/68071 |
Asiamoeba is a subgenus of the genus Plasmodium - all of which are parastic protozoa. The subgenus was created by Telford in 1988. Species in this subgenus infect lizards.
Diagnostic features[edit]
Species in the subgenus Asiamoeba have the following characteristics:
The schizonts and gametocytes are greatly disparate in size (4 to 15 times).
Species in this subgenus[edit]
References[edit]
| This infectious disease article is a stub. You can help Wikipedia by expanding it. |
| This chromalveolate article is a stub. You can help Wikipedia by expanding it. |
| License | http://creativecommons.org/licenses/by-sa/3.0/ |
| Rights holder/Author | Wikipedia |
| Source | http://en.wikipedia.org/w/index.php?title=Asiamoeba&oldid=574029716 |
Plasmodiid apicomplexa, causative agent of malaria, gamonts in erythrocytes; merogony in erythrocytes and other tissues depending on species; hemozoin deposited; vectors mosquitoes or phlebotomine flies; in mammals, birds, and reptiles; cause of estimated two million deaths annually; most important human species are P. vivax (causes benign tertian malaria) and P. falciparum (causesmalignant tertian malaria); Type species P. malariae
Bennettinia is a subgenus of the genus Plasmodium - all of which are parastic protozoa. The subgenus was created in 1997 by Valkiunas. [1]Species in this subgenus infect birds.
Diagnostic features[edit source | edit]
Species in the subgenus Bennettinia have the following characteristics:
Schizonts contain scant cytoplasm, are often round, do not exceed the size of the host nucleus and stick to it.
Gametocytes while varying in shape tend to be round or oval, do not exceed the size of the nucleus and stick to it.
Species in this subgenus[edit source | edit]
References[edit source | edit]
- ^ Valkiunas, G. (1997). Bird Haemosporidia. Institute of Ecology, Vilnius
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| This infectious disease article is a stub. You can help Wikipedia by expanding it. |
| This chromalveolate article is a stub. You can help Wikipedia by expanding it. |
| License | http://creativecommons.org/licenses/by-sa/3.0/ |
| Rights holder/Author | Wikipedia |
| Source | http://en.wikipedia.org/w/index.php?title=Bennettinia&oldid=568263564 |
Phylogeny based on Escalante et al., 1995; Perkins and Schall, 2002; Vargas-Serrato et al., 2003; Martinsen et al., 2008.
The field of Plasmodium phylogenetics has been particularly dynamic over the past decade, and continues to be so as more species are added to the tree and older hypotheses are called into question.
The study of the molecular phylogenetics of malaria parasites began in 1991 (Waters et al., 1991), and the field continues to grow. The conclusions of many earlier studies were overturned later, as they were affected by difficulties stemming from insufficient taxon sampling and problems with the genes chosen for analysis. The choice of both the ingroup taxa (the organisms of interest) and outgroup taxa (organisms outside the group of interest) for phylogenetic analysis can have a significant influence on the strength of the resulting evolutionary hypothesis. Older phylogenies often contained small numbers of ingroup species, between six and twelve, that were a mixture of very closely-related taxa and a few that were very divergent. This poor taxon sampling for this very large and diverse genus, coupled with the use of very distantly-related species as outgroups, probably lead to some spurious results and the conflicting conclusions found in the literature at the time.
In addition to problems of taxon sampling, for years the study of malaria systematics was also stymied by inherent problems in the loci chosen for analysis. Small subunit (SSU) rRNA and circumsporozoite protein (CSP) have been the workhorses of Plasmodium phylogenetics (Waters et al., 1991; Waters et al., 1993; Escalate and Ayala, 1994; Escalante et al., 1995; McCutchan, et al. 1996; Qari et al., 1996; Escalante et al., 1996; Escalante et al., 1997). Recent research indicates, however, that neither of these loci may be appropriate for evolutionary studies of Plasmodium species. SSU rRNA is a standard locus used in high-level molecular systematics, but it was later found that Plasmodium species possess separate genes of rRNA, each expressed at a different point in the life cycle, that are not evolving in a concerted manner and may still be exchanging genetic information with each other (Corredor and Enea, 1993). Several of the older phylogenies may have included a mixture of paralogs (duplicate genes) and orthologs (proper homologs), and only orthologs yield reliable gene trees. Although life-stage-specific primers have been developed for the SSU rRNA loci in Plasmodium, as gene conversion among the non-homogenized paralogs cannot be ruled out, this locus should be used with great caution. Indeed, even very recent phylogenies (Leclerc et al., 2004) using these sequences show results that cannot be reconciled with those of other loci in the genome.
Likewise, the locus CSP, which has been frequently used for evolutionary studies, may be problematic as the gene codes for a surface protein and is under strong selective pressure from the vertebrate immune system. Selectively driven, non-neutral changes in the gene may either obscure the phylogenetic signal or lead to incorrect phylogenetic inferences (Hughes and Hughes, 1995). CSP was a favored locus for study as large amounts of sequence data was available early on as the protein is of great interest to malaria science as a possible vaccine target. Improved methods for gene sequencing have allowed for better loci to be developed for evolutionary studies and CSP is now rarely used in systematics.
At this time, new loci are being developed for molecular studies, such as the mitochondrial gene for cytochrome b (Escalante et al., 1998; Perkins and Schall 2002) and the gene for the housekeeping enzyme adenylosuccinate lyase (Kedseierki et al., 2002). One caveat is that mitochondrial cytochrome b is the target of some antimalarial drugs, and mutations in the gene are known to be associated with resistance (see Vaidya, et al., 1993). Studies of these and other suitably chosen genes will hopefully lead to Plasmodium phylogenies based on the combined data from multiple loci. It should be noted, however, that the chance of identifying true neutrally evolving loci in Plasmodium species is compromised by the possibility of unrecognized selection pressure from the host immune system or (in the case of human parasites) drugs, and hence an extra measure of caution is called for.
One intuitive assumption that was challenged early on by molecular studies was the idea that human malaria parasites are closely related. Recent work now shows that, although primate parasites (with the exception of P. falciparum and P. reichenowi) grouped together, those parasites with a human host were not closely related within this group. The relatively distant relationships among human pathogens indicates that malaria most likely has an ancient association with primates (Escalante et al., 1998; Perkins and Schall, 2002; Leclerc et al., 2004, Cornejo and Escalante, 2006).
One of the most intriguing issues in malaria evolution is the mysterious origin of P. falciparum, and few issues have gone through as many significant revisions in the past 15 years. Older textbooks, and even some current ones, refer to P. falciparum as arising from a recent horizontal transfer from birds, and it is to this recent host shift that the high virulence of P. falciparum is often attributed (White, 2003). Newer research, however, refutes this hypothesis, as P. falciparum falls within a clade of mammalian parasites—distinct from and outside the group affecting birds and lizards. On the other hand, the species is also outside of the primate or rodent malarias. Rather, P. falciparum is in a divergent clade that includes only itself and its close sister species, the chimpanzee parasite, P. reichenowi.
The avian-origin hypothesis arose from some of the early molecular phylogenies that had problems due to insufficient taxon sampling and choice of genes (Waters et al., 1991; Waters et al., 1993; Escalante et al., 1995; Escalante et al., 1996; Escalante et al., 1997). First in 1996 (Qari et al., 1996) and then again in 2002 (Perkins and Schall, 2002), new evidence came to light refuting an avian-origin. Perkins and Schall (2002) published a relatively strong phylogeny based on cytochrome b with a large sample size of vertebrate parasites and an outgroup from a sister family. This cytochrome b phylogeny placed P. falciparum within the clade of mammalian parasites. In addition to the phylogenetics, this analysis included an explicit test of the avian-origin hypothesis using the Shimodaira-Hasegawa test, a phylogenetic tree-based method that calculates the likelihood of alternate trees, and rejected it.
At this time, the evidence (Qari et al., 1996; Escalate and Ayala, 1997; Perkins and Schall, 2002; Leclerc et al., 2004; Martinsen et al., 2008) indicates that the only species closely related to P. falciparum is P. reichenowi, and the two likely diverged from each other between 5 and 8 million years ago based on fossil dates of the human-chimpanzee split (Escalante and Ayala, 1994; Escalante et al., 1995).
Future phylogenetic analyses that include parasites that infect bats (several genera of malaria parasites have been described from these hosts alone) and ungulates may help to identify close relatives of P. falciparum and better understand the evolutionary history of these parasites.
| License | http://creativecommons.org/licenses/by-nc-sa/3.0/ |
| Rights holder/Author | Martine Zilversmit, Susan Perkins, Tree of Life web project |
| Source | http://tolweb.org/Plasmodium/68071 |
Sauramoeba is a subgenus of the genus Plasmodium - all of which are parastic protozoa. The subgenus was created in 1966 by Garnham.[1]Species in this subgenus infect reptiles.
Diagnostic features[edit]
The original criterion for inclusion in this genus was the presence of large schizonts giving rise to 12 or more merozoites. The criteria were subsequently revised by Telford in 1988.[2] The type species of this subgenus is Plasmodium diploglossi.
Species in the subgenus Sauramoeba have the following characteristics:
Large schizonts giving rise to 12 or more merozoites
The gametocytes like the schizonts are large.
Species in this subgenus[edit]
Plasmodium achiotense
Plasmodium adunyinkai
Plasmodium aeuminatum
Plasmodium agamae
Plasmodium beltrani
Plasmodium brumpti
Plasmodium cnemidophori
Plasmodium diploglossi
Plasmodium giganteum
Plasmodium heischi
Plasmodium josephinae
Plasmodium pelaezi
References[edit]
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| This medical article is a stub. You can help Wikipedia by expanding it. |
| This infectious disease article is a stub. You can help Wikipedia by expanding it. |
| This chromalveolate article is a stub. You can help Wikipedia by expanding it. |
| License | http://creativecommons.org/licenses/by-sa/3.0/ |
| Rights holder/Author | Wikipedia |
| Source | http://en.wikipedia.org/w/index.php?title=Sauramoeba&oldid=560910364 |
| This article needs additional citations for verification. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed. (November 2013) |
Plasmodium, commonly known as the malaria parasite, is a large genus of parasitic protozoa. As with some other genera of clinically important microorganisms, the genus name also yields a common noun; thus species of the genus are known as plasmodia. Infection with plasmodia is known as malaria, a deadly disease widespread in the tropics.
The parasite always has two hosts in its life cycle: a mosquito vector and a vertebrate host. The life-cycle is very complex, involving a sequence of different stages both in the vector and the host. These stages include sporozoites which are injected by the mosquito vector into the host's blood; latent hypnozoites which may rest undetected in the liver for up to 30 years; merosomes and merozoites which infect the red cells (erythrocytes) of the blood; trophozoites which grow in the red cells, and schizonts which divide there, producing more merozoites which leave to infect more red cells; and male and female sexual forms, gametocytes, which are taken up by other mosquitoes. In the mosquito's midgut, the gametocytes develop into gametes which fertilize each other to form motile zygotes which escape the gut, only to grow into new sporozoites which move to the mosquito's salivary glands, from where they are injected into the mosquito's next host, infecting it and restarting the cycle.
The genus Plasmodium was first described in 1885. It now contains about 200 species divided into several subgenera; as of 2006 the taxonomy was shifting, and species from other genera are likely to be added to Plasmodium. At least ten species infect humans; other species infect other animals, including birds, reptiles and rodents, while 29 species infect non-human primates. The parasite is thought to have originated from Dinoflagellates, photosynthetic protozoa.
The most common forms of human malaria are caused by Plasmodium falciparum, P. vivax, P. knowlesi, and P. malariae. P. falciparum, common in sub-Saharan Africa, and P. knowlesi, common in Southeast Asia, are especially dangerous.
Contents
§Taxonomy and host range[edit]
The genus Plasmodium was created in 1885 by Marchiafava and Celli and there are over 200 species recognized. New species continue to be described.[1]
As of 2006[update], the genus is in need of reorganization as it has been shown that parasites belonging to the genera Haemocystis and Hepatocystis appear to be closely related to Plasmodium. It is likely that other species such as Haemoproteus meleagridis will be included in this genus once it is revised.
Host range among the mammalian orders is non uniform. At least 29 species infect non-human primates; rodents outside the tropical parts of Africa are rarely affected; a few species are known to infect bats, porcupines and squirrels; carnivores, insectivores and marsupials are not known to act as hosts.
In 1898 Ronald Ross demonstrated the existence of Plasmodium in the wall of the midgut and salivary glands of a Culex mosquito. For this discovery he won the Nobel Prize in 1902. However credit must also be given to the Italian professor Giovanni Battista Grassi, who showed that human malaria could only be transmitted by Anopheles mosquitoes. For some species the vector may not be a mosquito.
Mosquitoes of the genera Culex, Anopheles, Culiseta, Mansonia and Aedes may act as vectors. The known vectors for human malaria (more than 100 species) belong to the genus Anopheles. Bird malaria is commonly carried by species belonging to the genus Culex. Only female mosquitoes bite. Aside from blood both sexes live on nectar, but one or more blood meals are needed by the female for egg laying, because there is very little protein in nectar.
§Life cycle[edit]

The life cycle of malaria parasites. A mosquito causes an infection by a bite. First, sporozoites enter the bloodstream, and migrate to the liver. They infect liver cells, where they multiply into merozoites, rupture the liver cells, and return to the bloodstream. Then, the merozoites infect red blood cells, where they develop into ring forms, trophozoites and schizonts that in turn produce further merozoites. Sexual forms are also produced, which, if taken up by a mosquito, will infect the insect and continue the life cycle.
The life cycle of Plasmodium is very complex. Sporozoites from the saliva of a biting female mosquito are transmitted to either the blood or the lymphatic system[2] of the recipient. The sporozoites then migrate to the liver and invade hepatocytes. This latent or dormant stage of the Plasmodium sporozoite in the liver is called the hypnozoite.
The development from the hepatic stages to the erythrocytic stages has been obscure. In 2006[3] it was shown that the parasite buds off the hepatocytes in merosomes containing hundreds or thousands of merozoites. These merosomes have been subsequently shown[4] to lodge in the pulmonary capillaries and to disintegrate there slowly over 48–72 hours releasing merozoites. Erythrocyte invasion is enhanced when blood flow is slow and the cells are tightly packed: both of these conditions are found in the alveolar capillaries.
Within the erythrocytes the merozoite grow first to a ring-shaped form and then to a larger trophozoite form. In the schizont stage, the parasite divides several times to produce new merozoites, which leave the red blood cells and travel within the bloodstream to invade new red blood cells. Most merozoites continue this replicative cycle, but some merozoites differentiate into male or female sexual forms (gametocytes) (also in the blood), which are taken up by the female mosquito.
In the mosquito's midgut, the gametocytes develop into gametes and fertilize each other, forming a zygote. After a brief period of inactivity, zygotes transform into a motile form called ookinetes. The ookinetes penetrate and escape the midgut, then embed themselves onto the exterior of the gut membrane and transform into oocysts. The nuclei of oocysts divides many times to produce large numbers of tiny elongated sporozoites. These sporozoites migrate to the salivary glands of the mosquito where they are injected into the blood of the next host the mosquito bites. The sporozoites move to the liver where they repeat the cycle.
The pattern of alternation of sexual and asexual reproduction is common in parasitic species. The evolutionary advantages of this type of life cycle were recognised by Mendel. Under favourable conditions, asexual reproduction is superior to sexual as the parent is well adapted to its environment and its descendents share all its genes. Transferring to a new host or in times of stress, sexual reproduction is generally superior as it shuffles the genes of two parents, producing a variety of individuals, some of which will be better adapted to the new environment.
Reactivation of the hypnozoites has been reported for up to 30 years after the initial infection in humans. The factors precipating this reactivation are not known. In the species Plasmodium malariae, Plasmodium ovale and Plasmodium vivax hypnozoites have been shown to occur. Reactivation does not occur in infections with Plasmodium falciparum. It is not known if hypnozoite reactivaction may occur with any of the remaining species that infect humans but this is presumed to be the case.
§Evolution[edit]
The life cycle of Plasmodium is best understood in terms of its evolution.
The Apicomplexa—the phylum to which Plasmodium belongs—are thought to have originated within the Dinoflagellates, a large group of photosynthetic protozoa. It is thought that the ancestors of the Apicomplexa were originally prey organisms that evolved the ability to invade the intestinal cells and subsequently lost their photosynthetic ability. Some extant Dinoflagellates, however, can invade the bodies of jellyfish and continue to photosynthesize, which is possible because jellyfish bodies are almost transparent. In other organisms with opaque bodies this ability would most likely rapidly be lost.
It is thought that Plasmodium evolved from a parasite spread by the fecal-oral route which infected the intestinal wall. At some point this parasite evolved the ability to infect the liver. This pattern is seen in the genus Cryptosporidium, to which Plasmodium is distantly related. At some later point this ancestor developed the ability to infect blood cells and to survive and infect mosquitoes. Plasmodium subsequently evolved a mechanism to invade the salivary glands of mosquitoes, allowing for transmission from mosquito to host. Once mosquito transmission was firmly established, the previous fecal-oral route was lost within the Plasmodium genus.[5]
The survivorship and relative fitness of mosquitoes are not adversely affected by Plasmodium infection which indicates the importance of vector fitness in shaping the evolution of Plasmodium.[6]Plasmodium has evolved the capability to manipulate mosquito feeding behavior. Mosquitoes harboring Plasmodium have a higher propensity to bite than uninfected mosquitoes. This tendency has facilitated the spread of Plasmodium to the various hosts. [7]
Current (2007) theory suggests that the genera Plasmodium, Hepatocystis and Haemoproteus evolved from Leukocytozoon species. Parasites of the genus Leukocytozoan infect white blood cells (leukocytes), liver and spleen cells and are transmitted by 'black flies' (Simulium species) — a large genus of flies related to the mosquitoes.
Leukocytes, hepatocytes and most spleen cells actively phagocytose particulate matter, making entry into the cell easier for the parasite. The mechanism of entry of Plasmodium species into erythrocytes is still very unclear, taking as it does less than 30 seconds. It is not yet known if this mechanism evolved before mosquitoes became the main vectors for transmission of Plasmodium.
Plasmodium evolved about 130 million years ago. This period coincided with the rapid spread of the angiosperms (flowering plants). This expansion in the angiosperms is thought to be due to at least one genomic duplication event. It seems probable that the increase in the number of flowers led to an increase in the number of mosquitoes and their contact with vertebrates.
Environmental factors play a considerable role in the evolution of Plasmodium and the transmission of malaria. The genetic information of Plasmodium falciparum has signaled a recent expansion that coincides with the agricultural revolution It is likely that the development of extensive agriculture increased mosquito population densities by giving rise to more breeding sites, which may have triggered the evolution and expansion of Plasmodium falciparum. [8]
Mosquitoes evolved in what is now South America about 230 million years ago. There are over 3500 species recognised but to date their evolution has not been well worked out so a number of gaps in our knowledge of the evolution of Plasmodium remain. It seems probable that birds were the first group infected by Plasmodium followed by the reptiles — probably the lizards. At some point primates and rodents became infected. The remaining species infected outside these groups seem likely to be due to relatively recent events.
There are over one hundred species of mosquito-transmitted Plasmodium. The phylogeny of these malarial parasites suggests that the Plasmodium of mammalian hosts forms a well-defined clade strongly associated with the specialization to the Anopheles mosquito vector. This was a major evolutionary transition that allowed Plasmodium to exploit humans and other mammals.[9]
P. falciparum, the most lethal malaria parasite of humans, evolved from a "nearly identical" parasite of western gorillas, not from chimpanzees, bonobos or ancient human populations.[10]
The high mortality and morbidity caused by malaria—especially that caused by P. falciparum —has placed the greatest selective pressure on the human genome in recent history. Several genetic factors provide some resistance to Plasmodium infection, including sickle cell trait, thalassaemia traits, glucose-6-phosphate dehydrogenase deficiency, and the absence of Duffy antigens on red blood cells.[11][12]
Although there are therapeutic medications to treat malaria, Plasmodium has accumulated increasing drug resistance over time. A recent examination has shown that even artemisinin, one of the most powerful anti-malarial drugs, has been experiencing decreased efficacy due to the development of resistance. [13]
§Molecular biology[edit]
All the species examined to date have 14 chromosomes, one mitochondrion and one plastid (also known as apicoplast, an organelle similar to a chloroplast). The chromosomes vary from 500 kilobases to 3.5 megabases in length. It is presumed that this is the pattern throughout the genus. The plastid, unlike those found in algae, is not photosynthetic. Its function is not fully known; however, it has been demonstrated that some essential metabolic pathways like isoprenoid, Fe-S clusters, fatty acid and phospholipid biosynthesis occurs in this organelle, and that it also still possess its own genome, partly shared with the nucleus.[14]
On a molecular level, the parasite damages red blood cells using plasmepsin enzymes — aspartic acid proteases which degrade hemoglobin.
§Taxonomy[edit]
Plasmodium belongs to the family Plasmodiidae (Levine, 1988), order Haemosporidia and phylum Apicomplexa. There are 450 recognised species in this order. Many species of this order are undergoing reexamination of their taxonomy with DNA analysis. It seems likely that many of these species will be reassigned after these studies have been completed.[15][16] For this reason the entire order is outlined here.
- Genus Plasmodium
- Subgenus Asiamoeba (lizards)
- Subgenus Bennettinia (birds)
- Subgenus Carinamoeba (reptiles)
- Subgenus Giovannolaia (birds)
- Subgenus Haemamoeba (birds)
- Subgenus Huffia (birds)
- Subgenus Lacertamoeba (reptiles)
- Subgenus Laverania (higher primates)
- Subgenus Novyella (birds)
- Subgenus Paraplasmodium (lizards)
- Subgenus Plasmodium (monkeys, higher primates)
- Subgenus Sauramoeba (reptiles)
- Subgenus Vinckeia (non-primate mammals)
- Genus Polychromophilus
- Genus Rayella
- Genus Saurocytozoon
The genera Plasmodium, Fallisia and Saurocytozoon all cause malaria in lizards. All are carried by Diptera (true two-winged flies). Pigment is absent in the Garnia. Non pigmented gametocytes are typically the only forms found in Saurocytozoon: pigmented forms may be found in the leukocytes occasionally. Fallisia produce non pigmented asexual and gametocyte forms in leukocytes and thrombocytes.
§Subgenera[edit]
The full taxonomic name of a species includes the subgenus but this is often omitted. The full name indicates some features of the morphology and type of host species.
The only two species in the sub genus Laverania are P. falciparum and P. reichenowi.
Species infecting monkeys and apes (the higher primates) with the exceptions of P. falciparum and P. reichenowi are classified in the subgenus Plasmodium.
Parasites infecting other mammals including lower primates (lemurs and others) are classified in the subgenus Vinckeia. The distinction between P. falciparum and P. reichenowi and the other species infecting higher primates was based on morphological findings but have since been confirmed by DNA analysis. Vinckeia, while previously considered to be something of a taxonomic 'rag bag', has been recently shown to form a coherent grouping. The remaining groupings here are based on the morphology of the parasites. Revisions to this system are likely as more species are subject to DNA analysis.
The four subgenera Giovannolaia, Haemamoeba, Huffia and Novyella were created by Corradetti et al.[17] for the known avian malarial species. A fifth — Bennettinia — was created in 1997 by Valkiunas.[18] The relationships between the subgenera are a matter of current investigation. Martinsen et al. 's recent (2006) paper outlines what was known at the time.[19]
As of 2007[update], P. juxtanucleare is the only known member of the subgenus Bennettinia.
Unlike the mammalian and bird malarias those affecting reptiles have been more difficult to classify. In 1966 Garnham classified those with large schizonts as Sauramoeba, those with small schizonts as Carinamoeba and the single then known species infecting snakes (Plasmodium wenyoni) as Ophidiella.[20] He was aware of the arbitrariness of this system and that it might not prove to be biologically valid. Telford in 1988 used this scheme as the basis for the accepted (2007) system.[21]
| Classification criteria |
|---|
| Avian species:
Species in the subgenus Bennettinia have the following characteristics:
Species in the subgenus Giovannolaia have the following characteristics:
Species in the subgenus Haemamoeba have the following characteristics:
Species in the subgenus Huffia have the following characteristics:
Species in the subgenus Novyella have the following characteristics:
Reptile species: Species in the subgenus Carinamoeba infect lizards. Their schizonts normally give rise to less than 8 merozoites, unlike those in the subgenus Sauramoeba which also infect lizards, but whose schizonts normally give rise to more than 8 merozoites.
Ophidiella was a subgenus created by Garnham in 1966 for the species infecting snakes. As of 2007[update] it was no longer in use. |
§Species infecting humans[edit]
The species of Plasmodium that infect humans include:
- Plasmodium falciparum (the cause of malignant tertian malaria)
- Plasmodium vivax (the most frequent cause of benign tertian malaria)
- Plasmodium ovale (the other, less frequent, cause of benign tertian malaria)
- Plasmodium malariae (the cause of benign quartan malaria)
- Plasmodium knowlesi (the cause of severe quotidian malaria in South East Asia since 1965)
- Plasmodium brasilianum
- Plasmodium cynomolgi
- Plasmodium cynomolgi bastianellii
- Plasmodium inui
- Plasmodium rhodiani
- Plasmodium schweitzi
- Plasmodium semiovale
- Plasmodium simium
The first four listed here are the most common species that infect humans. Nearly all human deaths from malaria are caused by the first species, P. falciparum, mainly in sub-Saharan Africa. With the use of the polymerase chain reaction additional species have been and are still being identified that infect humans.
One possible experimental infection has been reported with Plasmodium eylesi. Fever and low grade parasitemia were apparent at 15 days. The volunteer (Dr Bennett) had previously been infected by Plasmodium cynomolgi and the infection was not transferable to a gibbon (P. eylesi 's natural host) so this cannot be regarded as definitive evidence of its ability to infect humans. A second case has been reported that may have been a case of P. eylesi but the author was not certain of the infecting species.[22]
A possible infection with Plasmodium tenue has been reported.[23] This report described a case of malaria in a three-year-old black girl from Georgia, US, who had never been outside the US. She suffered from both P. falciparum and P. vivax malaria and while forms similar to those described for P. tenue were found in her blood even the author was skeptical about the validity of the diagnosis.
Confusingly, P. tenue was proposed in the same year (1914) for a species found in birds. The human species is now considered probably a misdiagnosis, and the bird species is described on the P. tenue page.
The only known host of P. falciparum and P. malariae is humans. P. vivax however can infect chimpanzees. Infection tends to be low grade but may be persistent and remain as source of parasites for humans for some time. P. vivax can also infect orangutans.[24]
P. ovale can be transmitted to chimpanzees. P. ovale has an unusual distribution, being found in Africa, the Philippines and New Guinea. In spite of its admittedly poor transmission to chimpanzees given its discontigous spread, it is suspected that P. ovale is a zoonosis with an as yet unidentified host. If so, the host is likely to be a primate. The remaining species capable of infecting humans all have other primate hosts.
Plasmodium shortii and Plasmodium osmaniae are now considered junior synonyms of Plasmodium inui
Taxonomy in parasitology before DNA based methods was always problematic, and revisions are continuing, leaving many obsolete names for Plasmodium species that infect humans.[25]
| Obsolete names for Plasmodium species infecting humans |
|---|
| P. camerense
P. causiasium |
§Infections in primates[edit]

Species of Plasmodium infect many primates across the world, such as the brown lemur, Eulemur fulvus, of Madagascar.
The species that infect primates other than humans include: P. bouillize, P. brasilianum, P. bucki, P. cercopitheci,P. coatneyi, P. coulangesi, P. cynomolgi, P. eylesi, P. fieldi, P. foleyi, P. fragile, P. girardi, P. georgesi, P. gonderi, P. hylobati, P. inui, P. jefferyi, P. joyeuxi, P. knowlesi, P. lemuris, P. percygarnhami, P. petersi, P. reichenowi, P. rodhaini, P. sandoshami, P. semnopitheci, P. silvaticum, P. simiovale, P. simium, P. uilenbergi, P. vivax and P. youngei.
Most if not all Plasmodium species infect more than one host: the host records shown here should be regarded as incomplete.
| Host records |
|---|
*P. bouillize — Cercopithecis campbelli
|
| Primate mosquito vectors |
|---|
*Anopheles albimanus — P. vivax
|
| Subspecies infecting primates |
|---|
*P. cynomolgi — P. cynomolgi bastianelli and P. cynomolgi ceylonensis.
The evolution of these species is still being worked out and the relationships given here should be regarded as tentative. This grouping, while originally made on morphological grounds, now has considerable support at the DNA level.
|
§Infections in non-primate mammals[edit]
The subgenus Vinckeia was created by Garnham to accommodate the mammalian parasites other than those infecting primates. Species infecting lemurs have also been included in this subgenus.
P. aegyptensis, P. bergei, P. chabaudi, P. inopinatum, P. yoelli and P. vinckei infect rodents. P. bergei, P. chabaudi, P. yoelli and P. vinckei have been used to study malarial infections in the laboratory. Other members of this subgenus infect other mammalian hosts.
| Host records |
|---|
*P. aegyptensis — Egyptian grass rat (Arvicanthis noloticus)[31]
|
| Vectors |
|---|
|
| Subspecies |
|---|
*P. berghei — P. berghei yoelii
Calomys callosus seems unlikely to be a natural host for P. atheruri, as P. atheruri is found in Africa and Calomys callosus in South America. |
| Less well documented species |
|---|
|
The species listed here from Courtney et al.[25] should be regarded as dubious.
P. epomophori of the bat (Hypsignathus monstruosus) has been reclassified as Hepatocystis epomophori. |
§Infections in birds[edit]

Many bird species, from raptors to passerines like the red-whiskered bulbul (Pycnonotus jocosus), can carry malaria.
Species in five Plasmodium subgenera infect birds — Bennettinia, Giovannolaia, Haemamoeba, Huffia and Novyella.[36]Giovannolaia appears to be a polyphyletic group and may be sudivided in the future.[19] DNA evidence is in 2014 helping to improve understanding of the diversity of Plasmodium species that infect birds.[37]
| Avian parasites |
|---|
| Species infecting birds include: P. accipiteris, P. alloelongatum, P. anasum, P. ashfordi, P. bambusicolai, P. bigueti, P. biziurae, P. buteonis, P. cathemerium, P. circumflexum, P. coggeshalli, P. corradettii, P. coturnix, P. dissanaikei, P. durae, P. elongatum, P. fallax, P forresteri, P. gallinacium, P. garnhami, P. giovannolai, P. griffithsi, P. gundersi, P. guangdong, P. hegneri, P. hermani, P. hexamerium, P. huffi, P. jiangi, P. juxtanucleare, P. kempi, P. lophurae, P.lutzi, P. matutinum, P. nucleophilum, P. papernai, P. paranucleophilum, P. parvulum, P. pediocetti, P. paddae, P. pinotti, P. polare, P. relictum, P. rouxi, P. tenue, P. tejerai, P. tumbayaensis and P. vaughani. |
| Avian host records |
|---|
*P. accipiteris — Levant sparrowhawk (Accipiter brevipes)
|
| Avian malaria vectors |
|---|
|
Aedes species:
Culex species:
Mansonia species:
|
| Avian malaria subspecies |
|---|
|
| Avian malaria inter-relatedness and doubtful species |
|---|
*P. durae is related to P. asanum, P. circumflexum, P. fallax, P. formosanum, P. gabaldoni, P. hegneri, P. lophrae, P. lophrae, P. pediocetti, P. pinotti, and P. polare.
A number of additional species have been described in birds — P. centropi, P. chloropsidis, P. gallinuae, P. herodialis, P. heroni, P. mornony, P. pericorcoti and P. ploceii — but the suggested speciation was based at least in part on the idea — 'one host — one species'. It has not been possible to reconcile the descriptions with any of the recognised species and these are not regarded as valid species. As further investigations are made into this genus these species may be resurrected. A species P. japonicum has been reported[48] but this appears to be the only report of this species and it should therefore be regarded of dubious validity. |
§Infections in reptiles[edit]

Over 3000 species of lizard, including the Carolina anole (Anolis carolinensis), carry some 90 kinds of malaria.
Species in the subgenera Asiamoeba, Carinamoeba, Lacertaemoba, Paraplasmodium and Sauramoeba infect reptiles.[49]
Over 90 species and subspecies of Plasmodium infect lizards and they have been reported from over 3200 species of lizard and 29 species of snake. Only three species — P. pessoai, P. tomodoni and P. wenyoni — infect snakes.
| Species infecting reptiles |
|---|
| P. achiotense, P. aeuminatum, P. agamae, P. arachniformis, P. attenuatum,P. aurulentum, P. australis, P. azurophilum, P. balli, P. basilisci, P. beebei, P. beltrani , P. brumpti, P. brygooi, P. chiricahuae, P. circularis, P. cnemaspi, P. cnemidophori, P. colombiense, P. cordyli, P. diminutivum, P. diploglossi, P. egerniae, P. fairchildi, P. floridense, P. gabaldoni, P. giganteum, P. gologoense, P. gracilis, P. guyannense, P. heischi, P. holaspi, P. icipeensis, P. iguanae, P. josephinae, P. kentropyxi, P. lacertiliae, P. lainsoni, P. lepidoptiformis, P. lionatum, P. loveridgei, P. lygosomae, P. mabuiae, P. mackerrasae, P. maculilabre, P. marginatum, P. mexicanum, P. michikoa, P. minasense, P. pelaezi, P. pessoai, P. pifanoi, P. pitmani, P. rhadinurum, P. sasai,P. saurocaudatum, P. scorzai, P. siamense, P. robinsoni, P. sasai, P. scorzai, P. tanzaniae, P. tomodoni, P. torrealbai, P. tribolonoti, P. tropiduri, P. uluguruense, P. uzungwiense, P. vacuolatum, P. vastator, P. volans, P. wenyoni and P. zonuriae. |
| Host records |
|---|
*P. agamae — the rainbow lizard (Agama agama)
|
| Vectors |
|---|
*P. agamae — Lutzomyia or Culicoides species
|
| Subspecies |
|---|
*P. fairchildi — P. fairchildi fairchildi and P. fairchildi hispaniolae
|
| Inter-relatedness |
|---|
| *P. floridense is closely related to P. tropiduri and P. minasense |
§Species reclassified into other genera[edit]
| Reclassified species |
|---|
As of 2007[update] the following species are regarded as belonging to the genus Hepatocystis rather than Plasmodium.
|
§References[edit]
- ^ Chavatte JM, Chiron F, Chabaud A, Landau I (March 2007). "[Probable speciations by "host-vector 'fidelity'": 14 species of Plasmodium from magpies]". Parasite (in French) 14 (1): 21–37. PMID 17432055.
- ^ "Malaria Parasites Develop in Lymph Nodes". HHMI News. Howard Hughes Medical Institute. 22 January 2006.
- ^ Sturm A, Amino R, van de Sand C, et al. (September 2006). "Manipulation of host hepatocytes by the malaria parasite for delivery into liver sinusoids". Science 313 (5791): 1287–90. doi:10.1126/science.1129720. PMID 16888102.
- ^ Baer K, Klotz C, Kappe SH, Schnieder T, Frevert U (November 2007). "Release of hepatic Plasmodium yoelii merozoites into the pulmonary microvasculature". PLoS Pathog. 3 (11): e171. doi:10.1371/journal.ppat.0030171. PMC 2065874. PMID 17997605.
- ^ Ghosh, A.K; Devenport, M; Jethwaney, D. "Malaria parasite invasion of the mosquito salivary glands requires interaction between the Plasmoidum TRAP and the Anopheles saglin proteins". PLoS Pathog. 5(1):e1000265.
- ^ Ferguson, HM; Mackinnon, MJ; Chan, BH; Read, AF. (2003). "Mosquito mortality and the evolution of malaria virulence". Evolution 57 (12): 2792–804. doi:10.1554/03-211.
- ^ Koella, J.C.; Sørensen, F.L.; Anderson, R.A. (1998). "The malaria parasite, Plasmodium falciparum, increases the frequency of multiple feeding of its mosquito vector, Anopheles gambiae". Proc Biol Sci 265 (1398): 763–8. doi:10.1098/rspb.1998.0358.
- ^ Hume J.C., Lyons E.J., Day K.P. 2003. Human migration, mosquitoes and the evolution of Plasmodium falciparum. Trends Parasitol. 19(3):144-9.
- ^ Martinsen ES, Perkins SL, Schall JJ. (2008). "A three-genome phylogeny of malaria parasites (Plasmodium and closely related genera): evolution of life-history traits and host switches". Molecular Phylogenetic Evolution 47 (1): 261–273. doi:10.1016/j.ympev.2007.11.012. PMID 18248741.
- ^ Weimin Liu, Yingying Li, Gerald H. Learn, Rebecca S. Rudicell, Joel D. Robertson, Brandon F. Keele, Jean-Bosco N. Ndjango, Crickette M. Sanz, David B. Morgan, Sabrina Locatelli, Mary K. Gonder, Philip J. Kranzusch, Peter D. Walsh, Eric Delaporte, Eitel Mpoudi-Ngole, Alexander V. Georgiev, Martin N. Muller, George M. Shaw, Martine Peeters, Paul M. Sharp, Julian C. Rayner & Beatrice H. Hahn (23 September 2010). "Origin of the human malaria parasite Plasmodium falciparum in gorillas" 467 (7314). pp. 420–425. doi:10.1038/nature09442. PMC 2997044. PMID 20864995.
- ^ Kwiatkowski DP (2005). "How malaria has affected the human genome and what human genetics can teach us about malaria". American Journal of Human Genetics 77 (2): 171–92. doi:10.1086/432519. PMC 1224522. PMID 16001361.

- ^ Hedrick PW (2011). "Population genetics of malaria resistance in humans". Heredity 107 (4): 283–304. doi:10.1038/hdy.2011.16. PMC 3182497. PMID 21427751.

- ^ Ashley, E.A.; Dhorda, M.; Fairhurst, R.M. et al. (2014). "Spread of artemisinin resistance in Plasmodium falciparum malaria". N Engl J Med 371 (5): 411–23. doi:10.1056/nejmoa1314981.
- ^ vanDooren, G; Striepen, B (2013). "The Algal Past and Parasite Present of Apicoplast". Annual Reviews of Microbiology 67: 271–289. doi:10.1146/annurev-micro-092412-155741. PMID 23808340.
- ^ Perkins SL, Schall JJ (October 2002). [0972:AMPOMP2.0.CO;2 "A molecular phylogeny of malarial parasites recovered from cytochrome b gene sequences"]. J. Parasitol. 88 (5): 972–8. doi:10.1645/0022-3395(2002)088[0972:AMPOMP]2.0.CO;2. PMID 12435139.
- ^ Yotoko, K.S.C.; Elisei C. (November 2006). "Malaria parasites (Apicomplexa, Haematozoea) and their relationships with their hosts: is there an evolutionary cost for the specialization?". J. Zoo. Syst. Evol. Res. 44 (4): 265–273. doi:10.1111/j.1439-0469.2006.00377.x.
- ^ Corradetti A., Garnham P.C.C., Laird M. (1963). "New classification of the avian malaria parasites". Parassitologia 5: 1–4.
- ^ Valkiunas G (1997). "Bird Haemosporidia". Acta Zoologica Lituanica 3–5: 1–607. ISSN 1392-1657.
- ^ a b c Martinsen ES, Waite JL, Schall JJ (April 2007). "Morphologically defined subgenera of Plasmodium from avian hosts: test of monophyly by phylogenetic analysis of two mitochondrial genes". Parasitology 134 (Pt 4): 483–90. doi:10.1017/S0031182006001922. PMID 17147839.
- ^ Garnham 1966
- ^ Telford S (1988). "A contribution to the systematics of the reptilian malaria parasites, family Plasmodiidae (Apicomplexa: Haemosporina)". Bulletin of the Florida State Museum Biological Sciences 34 (2): 65–96.
- ^ Tsukamoto M (1977). "An imported human malarial case characterized by severe multiple infections of the red blood cells". Ann. Trop. Med. Parasit. 19 (2): 95–104.
- ^ Russel P.F. (1928). "Plasmodium tenue (Stephens): A review of the literature and a case report". Am. J. Trop. Med. s1–8 (5): 449–479.
- ^ Reid MJ, Ursic R, Cooper D, et al. (December 2006). "Transmission of human and macaque Plasmodium spp. to ex-captive orangutans in Kalimantan, Indonesia". Emerging Infect. Dis. 12 (12): 1902–8. doi:10.3201/eid1212.060191. PMC 3291341. PMID 17326942.
- ^ a b Coatney G.R., Roudabush R.L. (1936). "A catalog and host-index of the genus Plasmodium". J. Parasitol. 22 (4): 338–353. doi:10.2307/3271859.
- ^ Collins WE, Sullivan JS, Nace D, Williams T, Williams A, Barnwell JW (February 2008). "Observations on the sporozoite transmission of Plasmodium vivax to monkeys". J. Parasitol. 94 (1): 287–8. doi:10.1645/GE-1283.1. PMID 18372652.
- ^ Collins WE, Richardson BB, Morris CL, Sullivan JS, Galland GG (July 1998). "Salvador II strain of Plasmodium vivax in Aotus monkeys and mosquitoes for transmission-blocking vaccine trials". Am. J. Trop. Med. Hyg. 59 (1): 29–34. PMID 9684622.
- ^ Collins WE, Sullivan JS, Nace D, et al. (April 2002). [0295:EIOAFW2.0.CO;2 "Experimental infection of Anopheles farauti with different species of Plasmodium"]. J. Parasitol. 88 (2): 295–8. doi:10.1645/0022-3395(2002)088[0295:EIOAFW]2.0.CO;2. PMID 12054000.
- ^ Collins WE, Morris CL, Richardson BB, Sullivan JS, Galland GG (August 1994). "Further studies on the sporozoite transmission of the Salvador I strain of Plasmodium vivax". J. Parasitol. 80 (4): 512–7. doi:10.2307/3283184. PMID 8064516.
- ^ Tan CH, Vythilingam I, Matusop A, Chan ST, Singh B (2008). "Bionomics of Anopheles latens in Kapit, Sarawak, Malaysian Borneo in relation to the transmission of zoonotic simian malaria parasite Plasmodium knowlesi". Malar. J. 7: 52. doi:10.1186/1475-2875-7-52. PMC 2292735. PMID 18377652.
- ^ Abd-el-Aziz GA, Landau I, Miltgen F (1975). "[Description of Plasmodium aegyptensis n. sp., presumed parasite of the Muridae Arvicanthis noloticus in Upper Egypt]". Ann Parasitol Hum Comp (in French) 50 (4): 419–24. PMID 1211772.
- ^ Sandosham AA, Yap LF, Omar I (September 1965). "A malaria parasite, plasmodium (Vinckeia) booliati sp.nov., from a Malayan giant flying squirrel". Med J Malaya 20 (1): 3–7. PMID 4221411.
- ^ Keymer IF (June 1966). "Studies on Plasmodium (Vinckeia) cephalophi of the grey duiker (Sylvicapra grimmia)". Ann Trop Med Parasitol 60 (2): 129–38. PMID 5962467.
- ^ Landau I, Chabaud AG (1978). "[Description of P. cyclopsi n. sp. a parasite of the microchiropteran bat Hipposideros cyclops in Gabon (author's transl)]". Ann Parasitol Hum Comp (in French) 53 (3): 247–53. PMID 697287.
- ^ Lien JC, Cross JH (December 1968). "Plasmodium (Vinckeia) watteni sp. n. from the Formosan giant flying squirrel, Petaurista petaurista grandis". J. Parasitol. 54 (6): 1171–4. doi:10.2307/3276986. PMID 5757690.
- ^ Wiersch SC, Maier WA, Kampen H (May 2005). "Plasmodium (Haemamoeba) cathemerium gene sequences for phylogenetic analysis of malaria parasites". Parasitol. Res. 96 (2): 90–4. doi:10.1007/s00436-005-1324-8. PMID 15812672.
- ^ Clark, Nicholas; Clegg, Sonya; Lima, Marcos (2014). "A review of global diversity in avian haemosporidians (Plasmodium and Haemoproteus: Haemosporida): new insights from molecular data". International Journal for Parasitology 44 (5): 329–338. doi:10.1016/j.ijpara.2014.01.004. PMID 24556563.
- ^ Valkiūnas G, Zehtindjiev P, Hellgren O, Ilieva M, Iezhova TA, Bensch S (May 2007). "Linkage between mitochondrial cytochrome b lineages and morphospecies of two avian malaria parasites, with a description of Plasmodium (Novyella) ashfordi sp. nov". Parasitol. Res. 100 (6): 1311–22. doi:10.1007/s00436-006-0409-3. PMID 17235548.
- ^ Landau I, Chabaud AG, Bertani S, Snounou G (December 2003). "Taxonomic status and re-description of Plasmodium relictum (Grassi et Feletti, 1891), Plasmodium maior Raffaele, 1931, and description of P. bigueti n. sp. in sparrows". Parassitologia 45 (3-4): 119–23. PMID 15267099.
- ^ Kirkpatrick CE, Lauer DM (January 1985). "Hematozoa of raptors from southern New Jersey and adjacent areas". J. Wildl. Dis. 21 (1): 1–6. doi:10.7589/0090-3558-21.1.1. PMID 3981737.
- ^ Earlé RA, Horak IG, Huchzermeyer FW, Bennett GF, Braack LE, Penzhorn BL (September 1991). "The prevalence of blood parasites in helmeted guineafowls, Numida meleagris, in the Kruger National Park". Onderstepoort J. Vet. Res. 58 (3): 145–7. PMID 1923376.
- ^ Valkiūnas G, Zehtindjiev P, Dimitrov D, Krizanauskiene A, Iezhova TA, Bensch S (May 2008). "Polymerase chain reaction-based identification of Plasmodium (Huffia) elongatum, with remarks on species identity of haemosporidian lineages deposited in GenBank". Parasitol. Res. 102 (6): 1185–93. doi:10.1007/s00436-008-0892-9. PMID 18270739.
- ^ Murata K, Nii R, Sasaki E, et al. (February 2008). "Plasmodium (Bennettinia) juxtanucleare infection in a captive white eared-pheasant (Crossoptilon crossoptilon) at a Japanese zoo". J. Vet. Med. Sci. 70 (2): 203–5. doi:10.1292/jvms.70.203. PMID 18319584.
- ^ Christensen BM, Barnes HJ, Rowley WA (July 1983). "Vertebrate host specificity and experimental vectors of Plasmodium (Novyella) kempi sp. n. from the eastern wild turkey in Iowa". J. Wildl. Dis. 19 (3): 204–13. doi:10.7589/0090-3558-19.3.204. PMID 6644918.
- ^ Manwell RD (November 1968). "Plasmodium octamerium n. sp., an avian malaria parasite from the pintail whydah bird Vidua macroura". J. Protozool. 15 (4): 680–5. doi:10.1111/j.1550-7408.1968.tb02194.x. PMID 5719065.
- ^ Valkiũnas G, Iezhova TA (August 2001). [0930:ACOTBP2.0.CO;2 "A comparison of the blood parasites in three subspecies of the yellow wagtail Motacilla flava"]. J. Parasitol. 87 (4): 930–4. doi:10.1645/0022-3395(2001)087[0930:ACOTBP]2.0.CO;2. PMID 11534666.
- ^ Poinar G (May 2005). "Plasmodium dominicana n. sp. (Plasmodiidae: Haemospororida) from Tertiary Dominican amber". Syst. Parasitol. 61 (1): 47–52. doi:10.1007/s11230-004-6354-6. PMID 15928991.
- ^ Manwell RD (February 1966). "Plasmodium japonicum, P. juxtanucleare and P. nucleophilum in the Far East". J. Protozool. 13 (1): 8–11. doi:10.1111/j.1550-7408.1966.tb01860.x. PMID 5912391.
- ^ Schall JJ (December 2000). "Transmission success of the malaria parasite Plasmodium mexicanum into its vector: role of gametocyte density and sex ratio". Parasitology 121 (Pt 6): 575–80. doi:10.1017/s0031182000006818. PMID 11155927.
- ^ Southgate BA (1970). "Plasmodium (Sauramoeba) giganteum in Agama cyanogaster: a new host record". Trans. R. Soc. Trop. Med. Hyg. 64 (1): 12–3. PMID 5462484.
- ^ Garnham PC, Telford SR (November 1984). "A new malaria parasite Plasmodium (Sauramoeba) heischi in skinks (Mabuya striata) from Nairobi, with a brief discussion of the distribution of malaria parasites in the family Scincidae". J. Protozool. 31 (4): 518–21. doi:10.1111/j.1550-7408.1984.tb05494.x. PMID 6512723.
- ^ Telford SR (October 1986). "Fallisia parasites (Haemosporidia: Plasmodiidae) from the flying lizard, Draco maculatus (Agamidae) in Thailand". J. Parasitol. 72 (5): 766–9. doi:10.2307/3281471. PMID 3100759.
- ^ a b Telford SR (1979). "A taxonomic revision of small neotropical saurian Malarias allied to Plasmodium minasense". Ann Parasitol Hum Comp 54 (4): 409–22. PMID 533109.
- ^ Telford SR, Telford SR (April 2003). [0362:RAROPP2.0.CO;2 "Rediscovery and redescription of Plasmodium pifanoi and description of two additional Plasmodium parasites of Venezuelan lizards"]. J. Parasitol. 89 (2): 362–8. doi:10.1645/0022-3395(2003)089[0362:RAROPP]2.0.CO;2. PMID 12760655.
§Further reading[edit]
§Identification[edit]
- Garnham, P.C.C. (1966). Malaria Parasites And Other Haemosporidia. Oxford: Blackwell. ISBN 0397601328.
- Hewitt, R.I. (1940). Bird Malaria. American Journal of Hygiene 15. Baltimore: Johns Hopkins Press.
- Laird, M. (1998). Avian Malaria in the Asian Tropical Subregion. Singapore: Springer. ISBN 9813083190.
§Biology[edit]
- Baldacci P, Ménard R (October 2004). "The elusive malaria sporozoite in the mammalian host". Mol. Microbiol. 54 (2): 298–306. doi:10.1111/j.1365-2958.2004.04275.x. PMID 15469504.
- Bledsoe GH (December 2005). "Malaria primer for clinicians in the United States" (PDF). South. Med. J. 98 (12): 1197–204; quiz 1205, 1230. doi:10.1097/01.smj.0000189904.50838.eb. PMID 16440920.
- Shortt HE (1951). "Life-cycle of the mammalian malaria parasite". Br. Med. Bull. 8 (1): 7–9. PMID 14944807.
§History[edit]
- Slater LB (2005). "Malarial birds: modeling infectious human disease in animals". Bull Hist Med 79 (2): 261–94. doi:10.1353/bhm.2005.0092. PMID 15965289.
§External links[edit]
| Wikispecies has information related to: Plasmodium |
- Malaria Atlas Project
- "Plasmodium". NCBI Taxonomy Browser. 5820.
| License | http://creativecommons.org/licenses/by-sa/3.0/ |
| Rights holder/Author | Wikipedia |
| Source | http://en.wikipedia.org/w/index.php?title=Plasmodium&oldid=650322698 |
Plasmodium incertae is a parasite of the genus Plasmodium subgenus Vinckeia.
Like all Plasmodium species P. incertae has both vertebrate and insect hosts. The vertebrate hosts for this parasite are mammals.
Contents |
Geographical occurrence
This species occurs in Asia.
Vectors
Not known.
Clinical features and host pathology
This species infects Asian flying squirrels.
References
| This Plasmodium-related article is a stub. You can help Wikipedia by expanding it. |
| License | http://creativecommons.org/licenses/by-sa/3.0/ |
| Rights holder/Author | Wikipedia |
| Source | http://en.wikipedia.org/w/index.php?title=Plasmodium_incertae&oldid=545503496 |
|
|
|
The Plasmodium species infecting primates include the parasites causing malaria in humans.
Contents
- 1 Species infecting humans
- 2 Plasmodium taxonomy
- 3 Species previously described as infecting humans but no longer recognised as valid
- 4 Species infecting other hosts
- 5 Primate groups and Plasmodium species
- 6 Mosquitoes known to transmit human malaria listed by region
- 7 Primate mosquito vectors and associated Plasmodium species
- 8 References
Species infecting humans[edit]
Common infections[edit]
- Plasmodium falciparum (the cause of malignant tertian malaria)
- Plasmodium vivax (the most frequent cause of benign tertian malaria)
- Plasmodium ovale curtisi (another, less frequent, cause of benign tertian malaria)
- Plasmodium ovale wallikeri (another, less frequent, cause of benign tertian malaria)
- Plasmodium malariae (the cause of benign quartan malaria)
- Plasmodium knowlesi (the cause of severe quotidian malaria in Southeast Asia)
Rare cases[edit]
While infection of humans by other species is known, they are quite rare, in some instances, only a single case. In a number of the cases, the means of infection is unknown, and may be due to accident, i.e. infection by laboratory equipment or a bite by an animal. With the use of the polymerase chain reaction additional species have been and are still being identified that infect humans.
- Plasmodium cynomolgi (spp. cynomolgi, bastianellii)
(Plasmodium brasilianum and Plasmodium rhodiani which have been reported to infect humans, are likely synonymous with P. malariae)
One possible experimental infection has been reported with Plasmodium eylesi. Fever and low grade parasitemia were apparent at 15 days. The volunteer (Dr Bennett) had previously been infected by Plasmodium cynomolgi and the infection was not transferable to a gibbon (P. eylesi 's natural host) so this cannot be regarded as definitive evidence of its ability to infect humans. A second case has been reported that may have been a case of P. eylesi but the author was not certain of the infecting species.[4]
A possible infection with Plasmodium tenue has been reported.[5] This report described a case of malaria in a three-year-old black girl from Georgia, USA who had never been outside the US. She suffered from both P. falciparum and P. vivax malaria and while forms similar to those described for P. tenue were found in her blood even the author was skeptical about the validity of the diagnosis.
Confusingly Plasmodium tenue was proposed in the same year (1914) for a species found in birds. The human species is now considered to be likely to have been a misdiagnosis and the bird species is described on the Plasmodium tenue page.
Notes[edit]
- falciparum
Until recently the only known host of P. falciparum was humans but this species has also been described in gorillas (Gorilla gorilla)[6] and bonobos[7] There has been a single report of P. falciparum in a brown howler monkey (Alouatta guariba) and in black howler monkeys (Alouatta caraya)[8] but until this is confirmed its validity should be considered dubious.
A possible report of P. falciparum in a greater spot-nosed monkeys (Cercopithecus nictitans) has not been confirmed in a large survey.[9]
A species that clusters with P. falciparum and P. reichenowi has been identified in Gabon, Africa in chimpanzees (Pan troglodytes).[10] This appears to have diverged from these two species about 21 million years ago. It has only been identified from the sequence of its mitochondrion to date and further work is needed to characterise the species. A second report has confirmed the existence of this species in chimpanzees.[6] A third report has confirmed the existence of this species.[11]
Night monkeys (Aotus nigriceps) can be infected with P. falciparum. This infection may occur naturally.[12] Their potential role - if any - as a source of human infection is unknown.
Two additional species within the subgenus Laverania have been identified on the basis of DNA sequences alone: Plasmodium billbrayi and Plasmodium billcollinsi.[6] and bonobos[7]P. billbrayi was found in two subspecies of chimpanzee (Pan troglodytes troglodytes and Pan troglodytes schweinfurthii). P. billcollinsi was found in only one subspecies of chimpanzee (Pan troglodytes troglodytes). Further work is needed to characterise these species.
- malariae
Humans are currently considered to be the only host for P. malariae. However Rodhain and Dellaert in the 1940s showed with transmission studies that P. malariae was present in chimpanzees.[13][14] The presence of P. malaria in chimpanzees has been reported in Japan suggesting that this species may be able to act as a host.[15] A second paper has described the presence of P. malaria in wild chimpanzees.[11] Another paper has reported several cases of P. malariae in brown howler monkey (Alouatta guariba) and black howler monkeys (Alouatta caraya)[8] It has been shown that splectomised three-striped night monkey (Aotus trivirgatus) can be infected with P. malariae.[16] Another paper has confirmed the presence of P. malaria in chimpanzees.[17]
The existence of multiple independent reports seem to suggest that the chimpanzee and possibly other species may act as a host to P. malaria at least occasionally.
- vivax
P. vivax will infect chimpanzees. Infection tends to be low grade but may be persistent and remain as source of parasites for humans for some time. P. vivax is also known to infect orangutans[18] and the brown howler monkey (Alouatta guariba clamitans)[8]P. vivax has been reported from chimpanzees living in the wild.[11] It has been suggested that vivax infection of the great apes in Africa may act as a reservoir given the prevalence of Duffy antigen negative humans in this area.[19]
- ovale
Like P. vivax, P. ovale has been shown to be transmittable to chimpanzees. P. ovale has an unusual distribution pattern being found in Africa, Myanmar the Philippines and New Guinea. In spite of its admittedly poor transmission to chimpanzees given its discontigous spread, it is suspected that P. ovale may in fact be a zooenosis with an as yet unidentified host. If this is actually the case, the host seems likely to be a primate. A report has been published suggesting that P. ovale may be a natural parasite of chimpanzees[20] but this needs confirmation. P. ovale has since been described from chimpanzees living in the wild.[11] This suggests that human infection with this species may as previously suspected be a zoonosis.
It has been recently shown that P. ovale is actually two genetically distinct species that coexist. These species are Plasmodium ovale curtisi and Plasmodium ovale wallikeri.[21] These two species separated between 1.0 and 3.5 million years ago.
- knowlesi
Plasmodium knowlesi has a natural reservoir in the macaques of Southeast Asia, and was only in 1965 identified as being transmissible to humans.
- Other species
The remaining species capable of infecting humans all have other primate hosts.
Plasmodium taxonomy[edit]
- P. cynomolgi - P. cynomolgi bastianelli, P. cynomolgi ceylonensis and P. cynomolgi cynomolgi.
- P. inui - P. inui inui and P. inui shortii
- P. knowlesi - P. knowlesi edesoni and P. knowlesi knowlesi.
- P. ovale - P. ovale curtisi and P. ovale wallikeri
- P. vivax - P. vivax hibernans, P. vivax chesson and P. vivax multinucleatum.
Interrelatedness - The evolution of these species is still being worked out and the relationships given here should be regarded as tentative. This grouping, while originally made on morphological grounds, now has considerable support at the DNA level.
- P. brasilianum, P. inui and P. rodhaini are similar to P. malariae (quartan malaria group)
- P. cynomolgi, P. fragile, P. knowlesi, P. simium and P. schwetzi are similar to P. vivax
- P. fieldi and P. simiovale are similar to P. ovale
- P. falciparum is closely related to P. reichenowi.
Notes[edit]
- P. kochi has been described as a parasite of monkeys. This species is currently classified as Hepatocystis kochi. This may be subject to revision.
- P. brasilianum and P. rodhaini seem likely to be the same species as P. malariae.
- P. lemuris may actually belong to the Haemoproteus genus. Clarification of this point awaits DNA examination.
- P. shortii is currently (2007) regarded as a junior synonym of P. inui.
Species previously described as infecting humans but no longer recognised as valid[edit]
Taxonomy in parasitology until the advent of DNA based methods has always been a problem and revisions in this area are continuing. A number of synonyms have been given for the species infecting humans that are no longer recognised as valid.[22] Since perusal of the older literature may be confusing some currently defunct species names are listed here.
P. causiasium
P. golgi
P. immaculatum
P. laverani var. tertium
P. laverani var. quartum
P. malariae var. immaculatum
P. malariae var. incolor
P. malariae var. irregularis
P. malariae var. parva
P. malariae var. quartanae
P. malariae var. quotidianae
P. perniciosum
P. pleurodyniae
P. praecox
P. quartana
P. quotidianum
P. sedecimanae
P. tenue
P. undecimanae
P. vegesio-tertaniae
P. vivax-minuta
Plasmodium shortii and Plasmodium osmaniae are now considered to be junior synonyms of Plasmodium inui.
Species infecting other hosts[edit]
|
|
This section contains an enumeration of examples, but lacks a general overview of its topic. You can help by adding an appropriate introductory section. Editing help is available. (March 2014) |
Most if not all Plasmodium species infect more than one host: the host records shown here should be regarded as incomplete.
- P. billbrayi - chimpanzees (Pan troglodytes troglodytes, Pan troglodytes schweinfurthii)
- P. billcollinsi - chimpanzees (Pan troglodytes troglodytes, Pan troglodytes schweinfurthii)
- P. bouillize - Cercopithecis campbelli
- P. brasilianum - Alouatta fusca, Alouatta palliata, Alouatta seniculus straminea, Alouatta villosa, several night monkey (Aotus) species, Aotus nigriceps, Ateles fusciceps, Ateles geoffroyi, Ateles geoffroyi grisescens, Ateles paniscus, Ateles paniscus paniscus, Ateles paniscus chamek, Brachyteles arachnoides, Callicebus moloch ornatus, Callicebus torquatus, Cebus albifrons, Cebus apella, Cebus capucinus, Cebus capucinus capucinus, Cebus capucinus imitator, Chiropotes chiropotes, Lagothrix cana, Lagothrix infumata, Lagothrix lagotricha, Saimiri boliviense, Saimiri sciureus and Saimiri ustus.
- P. bucki - Lemur macaco macaco
- P. cercopitheci - Cercopithecis nictitans
- P. coatneyi - crab eating macque (Macaca fascicularis) and Javanese long-tailed macaque (Macaca irus), silvered leaf monkey (Presbytis cristatus)
- P. coulangesi - Lemur macaco macaco
- P. cynomolgi - bear macaque (Macaca arctoides), Formosan rock macaque (Macaca cyclopis), crab eating macque (Macaca fascicularis), Javanese long-tailed macaque (Macaca irus), Rhesus monkey (Macaca mulatta), southern pig-tailed macaque (Macaca nemestrina), bonnet macaque (Macaca radiata), toque macaque (Macaca sinica), orangutan (Pongo species), silver leaf monkey (Presbytis cristatus) and Hanuman langur (Presbytis entellus)
- P. eylesi - several gibbon (Hylobates) species including Hylobates lar
- P. falciparum - gorillas (Gorilla gorilla), bonobos (Pan paniscus)
- P. fieldi - the crab eating macque (Macaca fascicularis), the Rhesus monkey (Macaca mulatta), the pig-tailed macaque (Macaca nemestrina), the bonnet macaque (Macaca radiata) and the baboon (Papio doguera).
- P. foleyi - Lemur fulvus rufus
- P. fragile - Aotus lemurinus griseimembra, Aotus nancymaae, Aotus vociferans, crab eating macque (Macaca fascicularis), Rhesus monkey (Macaca mulatta), bonnet macaque (Macaca radiata), toque macaque (Macaca sinica) and Saimiri boliviensis
- P. inui - Formosan rock macaque (Macaca cyclopis), crab eating macque (Macaca fascicularis) and Javanese long-tailed macaque (Macaca irus)
- P. gaboni - chimpanzees (Pan troglodytes)[11]
- P. georgesi - black crested mangabey (Cercocebus albigena)
- P. girardi - Lemur fulvus rufus, Lemur macaco macaco
- P. gonderi - black crested mangabey (Cercocebus albigena), Cercocebus aterrimus, sooty mangabey (Cercocebus atys), Tana River mangabey (Cercocebus galeritus agilus), crab eating macque (Macaca fascicularis) and drills (Mandrillus leucophaeus)
- P. gora - gorillas (Gorilla gorilla)
- P. gorb - gorillas (Gorilla gorilla)
- P. hylobati - several gibbon (Hylobates) species including Hylobates lar and Hylobates moloch
- P. inui - Aotus lemurinus griseimembra, Aotus nancymaae, Celebes black ape (Cynopithecus niger), Assamese macques (Macaca assamensis), crab eating macque (Macaca fascicularis), Rhesus monkey (Macaca mulatta), southern pig-tailed macaque (Macaca nemestrina), bonnet macaque (Macaca radiata), several Presbytis species, Saimiri boliviensis
- P. joyeuxi - Cercopithecis callitricus
- P. knowlesi - crab eating macque (Macaca fascicularis), pig-tailed macaque (Macaca nemestrina) and Presbytis malalophus
- P. knowlesi edesoni - Javanese long-tailed macaque (Macaca irus)
- P. lemuris - lemurs (Lemur collaris, Lemur macaco macaco)
- P. malagasi - lemurs
- P. malariae - brown howler monkey (Alouatta guariba clamitans), black howler monkey (Alouatta caraya) chimpanzee (')[11]
- P. percygarnhami - Lemur macaco macaco
- P. petersi - black crested mangabey (Cercocebus albigena)
- P. pitheci - orangutans (Pongo pygmaeus)
- P. sandoshami - Malayan flying lemur (Cynocephalus variegatus)
- P. semnopitheci - northern plains gray langur (Semnopithecus entellus)
- P. semiovale - toque macaque (Macaca sinica)
- P. shortii - bonnet macaque (Macaca radiata) and toque macaque (Macaca sinica)
- P. silvaticum - orangutans (Pongo pygmaeus)
- P. simium - several howler monkeys (Alouatta) species including the brown howler monkey (Alouatta fusca) and woolly spider monkey (Brachyteles arachnoides)
- P. uilenbergi - Lemur fulvus fulvus
- P. vivax - orangutans (Pongo species), chimpanzees (Pan),[11] monkeys Saimiri boliviensis,[23]Aotus lemurinus griseimambra,[24] the brown howler monkey (Alouatta guariba clamitans) and tamarins (Saguinus mystax and Saguinus fuscicollis)
- P. youngei - white handed gibbon (Hylobates lar)
It has been proposed that the species P. gora and P. gorb should be renamed P. adleri and P. blacklocki respectively.
Primate groups and Plasmodium species[edit]
New World monkeys of the family Cebidae: P. brasilianum and P. simium
Old World monkeys of the Cercopithecidae family: P. coatneyi, P. cynomolgi, P. fieldi, P. fragile, P.gonderi, P. georgesi, P. inui, P. knowlesi, P. petersi, P. shortti and P. simiovale
Gibbons of the Hylobatidae family: P. eylesi, P. hylobati, P. jefferyi and P. youngi
Orangutans (Pongo): P. pitheci and P. silvaticum
Gorillas and chimpanzees: P. billcollini, P. billbrayii, P. falciparum, P. gabonensi, P. gora, P. gorb, P. reichenowi, P. rodhaini and P. schwetzi
Mosquitoes known to transmit human malaria listed by region[edit]
|
|
This section contains an enumeration of examples, but lacks a general overview of its topic. You can help by adding an appropriate introductory section. Editing help is available. (March 2014) |
This listing may be incomplete as the taxonomy of this genus is under revision.
North American
- Anopheles (Anopheles) freeborni
- Anopheles (Anopheles) quadrimaculatus
- Anopheles (Nyssorhynchus) albimarus
Central American
- Anopheles (Anopheles) aztecus
- Anopheles (Anopheles) punctimacula
- Anopheles (Anopheles) pseudopunctipennis
- Anopheles (Nyssorhynchus) albimanus
- Anopheles (Nyssorhynchus) albitarsis
- Anopheles (Nyssorhynchus) aquasalis
- Anopheles (Nyssorhynchus) argyritarsis
- Anopheles (Nyssorhynchus) darlingi
South American
- Anopheles (Anopheles) pseudopunctipennis
- Anopheles (Anopheles) punctimacula
- Anopheles (Kerteszia) bellator
- Anopheles (Kerteszia) cruzii
- Anopheles (Kerteszia) neivai
- Anopheles (Nyssorhynchus) albimanus
- Anopheles (Nyssorhynchus) albitarsis
- Anopheles (Nyssorhynchus) aquasalis
- Anopheles (Nyssorhynchus) argyritarsis
- Anopheles (Nyssorhynchus) braziliensis
- Anopheles (Nyssorhynchus) darlingi
- Anopheles (Nyssorhynchus) nuneztovari
- Anopheles (Nyssorhynchus) triannulatus
North Eurasian
- Anopheles (Anopheles) atroparvus
- Anopheles (Anopheles) messeae
- Anopheles (Anopheles) sacharovi
- Anopheles (Anopheles) sinensis
- Anopheles (Cellia) pattoni
Mediterranean
- Anopheles (Anopheles) atroparvus
- Anopheles (Anopheles) claviger
- Anopheles (Anopheles) labranchiae
- Anopheles (Anopheles) messeae
- Anopheles (Anopheles) sacharovi
- Anopheles (Cellia) Hispaniola
- Anopheles (Cellia) superpictus
Afro-Arabian
- Anopheles (Cellia) culicifacies
- Anopheles (Cellia) fluviatilis
- Anopheles (Cellia) Hispaniola
- Anopheles (Cellia) multicolor
- Anopheles (Cellia) pharoensis
- Anopheles (Cellia) sergentii
Afrotropical
- Anopheles (Cellia) arabiensis
- Anopheles (Cellia) funestus
- Anopheles (Cellia) gambiae
- Anopheles (Cellia) melas
- Anopheles (Cellia) merus
- Anopheles (Cellia) moucheti
- Anopheles (Cellia) nili
- Anopheles (Cellia) pharoensis
Indo-Iranian
- Anopheles (Anopheles) sacharovi
- Anopheles (Cellia) aconitus
- Anopheles (Cellia) annularis
- Anopheles (Cellia) culicifacies
- Anopheles (Cellia) fluviatilis
- Anopheles (Cellia) jeyporiensis
- Anopheles (Cellia) minimus
- Anopheles (Cellia) philippinensis
- Anopheles (Cellia) pulcherrimus
- Anopheles (Cellia) stephensi
- Anopheles (Cellia) sundaicus
- Anopheles (Cellia) superpictus
- Anopheles (Cellia) tessellatus
- Anopheles (Cellia) varuna
Indo-Chinese hills
- Anopheles (Anopheles) nigerrimus
- Anopheles (Cellia) annularis
- Anopheles (Cellia) culicifacies
- Anopheles (Cellia) dirus
- Anopheles (Cellia) fluviatilis
- Anopheles (Cellia) jeyporiensis
- Anopheles (Cellia) maculatus
- Anopheles (Cellia) minimus
Malaysian
- Anopheles (Anopheles) campestris
- Anopheles (Anopheles) conaldi
- Anopheles (Anopheles) donaldi
- Anopheles (Anopheles) letifer
- Anopheles (Anopheles) nigerrimus
- Anopheles (Anopheles) whartoni
- Anopheles (Cellia) acconitus
- Anopheles (Cellia) balabacensis
- Anopheles (Cellia) dirus
- Anopheles (Cellia) flavirostris
- Anopheles (Cellia) jeyporiensis
- Anopheles (Cellia) leucosphyrus
- Anopheles (Cellia) ludlowae
- Anopheles (Cellia) maculatus
- Anopheles (Cellia) mangyanu
- Anopheles (Cellia) minimus
- Anopheles (Cellia) philippiensis
- Anopheles (Cellia) subpictus
- Anopheles (Cellia) sundaicus
Chinese
- Anopheles (Anopheles) anthropophagus
- Anopheles (Anopheles) sinensis
- Anopheles (Cellia) balabacensis
- Anopheles (Cellia) jeyporiensis
- Anopheles (Cellia) pattoni
Australasian
- Anopheles (Anopheles) bacroftii
- Anopheles (Cellia) farauti type 1
- Anopheles (Cellia) farauti type 2
- Anopheles (Cellia) hilli
- Anopheles (Cellia) karwari
- Anopheles (Cellia) koliensis
- Anopheles (Cellia) punctulatus
- Anopheles (Cellia) subpictus
Primate mosquito vectors and associated Plasmodium species[edit]
|
|
This section contains an enumeration of examples, but lacks a general overview of its topic. You can help by adding an appropriate introductory section. Editing help is available. (March 2014) |
- Anopheles (Nyssorhynchus) albimanus - P. fieldi, P. vivax
- Anopheles (Nyssorhynchus) albitarsis - P. vivax
- Anopheles (Nyssorhynchus) aquasalis - P. vivax
- Anopheles (Cellia) arabensis - P. falciparum
- Anopheles (Nyssorhynchus) argyritarsi - P. vivax
- Anopheles (Anopheles) argyropus - P. fieldi
- Anopheles (Anopheles) artemievi - P. vivax
- Anopheles (Anopheles) atroparvus - P. fieldi, P. vivax
- Anopheles (Anopheles) aztecus - P. vivax
- Anopheles (Cellia) baimaii - P. vivax
- Anopheles (Cellia) balabacensis - P. fieldi, P. vivax
- Anopheles (Anopheles) beklemishevi - P. vivax
- Anopheles (Kerteszia) bellator - P. vivax
- Anopheles (Nyssorhynchus) benarrochi - P. vivax
- Anopheles (Kertezia) bifurcatus - P. vivax
- Anopheles (Nyssorhynchus) braziliensis - P. vivax
- Anopheles (Kertezia) claviger - P. vivax
- Anopheles (Anopheles) coustani - P. falciparum
- Anopheles (Kerteszia) cruzii - P. vivax
- Anopheles (Cellia) culicifacies - P. vivax
- Anopheles (Nyssorhynchus) darlingi - P. falciparum, P. vivax
- Anopheles (Nyssorhynchus) deaneorum - P. falciparum, P. vivax
- Anopheles (Cellia) dirus - P. cynomolgi, P. fieldi, P. falciparum, P. inui, P. vivax
- Anopheles (Anopheles) donaldi - P. fieldi
- Anopheles (Nyssorhynchus) dunhami - P. vivax
- Anopheles (Cellia) epiroticus - P. vivax
- Anopheles (Cellia) farauti - P. coatneyi, P. vivax[25]
- Anopheles (Cellia) flavirostris - P. vivax
- Anopheles (Anopheles) freeborni - P. fieldi, P. vivax[26]
- Anopheles (Cellia) funestus - P. falciparum
- Anopheles (Cellia) fluviatilis - P. vivax
- Anopheles (Cellia) gambiae - P. falciparum, P. vivax
- Anopheles (Cellia) hackeri - P. fieldi, P. knowlesi[27]
- Anopheles (Kerteszia) homunculus - P. vivax
- Anopheles (Anopheles) hyrcanus - P. vivax
- Anopheles (Cellia) introlatus - P. cynomolgi, P. eylesi
- Anopheles (Anopheles) kleini - P. vivax
- Anopheles (Cellia) kochi - P. eylesi, P. fieldi
- Anopheles (Cellia) latens - P. knowlesi[28][29]
- Anopheles (Anopheles) lesteri - P. eylesi
- Anopheles (Anopheles) letifer - P. eylesi, P. fieldi
- Anopheles (Cellia) leucosphyrus - P. eylesi, P. vivax
- Anopheles (Cellia) maculatus - P. eylesi, P. fieldi, P. inui, P. vivax, P. youngei
- Anopheles (Nyssorhynchus) marajoara - P. vivax
- Anopheles (Anopheles) maculipennis - P. vivax
- Anopheles (Anopheles) martinius - P. vivax
- Anopheles (Anopheles) mediopunctatus - P. falciparum, P. vivax
- Anopheles (Cellia) melas - P. falciparum
- Anopheles (Cellia) merus - P. falciparum
- Anopheles (Anopheles) messeae - P. vivax
- Anopheles (Cellia) minimus - P. vivax
- Anopheles (Cellia) moucheti - P. falciparum
- Anopheles (Cellia) nili - P. falciparum
- Anopheles (Nyssorhynchus) nuneztovari - P. vivax
- Anopheles (Nyssorhynchus) oswaldoi - P. falciparum, P. vivax
- Anopheles (Anopheles) paludis - P. falciparum
- Anopheles (Anopheles) peditaeniatus - P. fieldi
- Anopheles (Cellia) philippinensis - P. fieldi
- Anopheles (Anopheles) pseudopunctipennis - P. vivax
- Anopheles (Cellia) pulcherrimus - P. vivax
- Anopheles (Anopheles) pullus - P. vivax
- Anopheles (Anopheles) punctimacula - P. vivax
- Anopheles (Anopheles) punctipennis - P. vivax
- Anopheles (Cellia) quadrimaculatus - P. fieldi, P. vivax
- Anopheles (Nyssorhynchus) rangeli - P. vivax
- Anopheles (Cellia) macarthuri - P. eylesi
- Anopheles (Anopheles) roperi - P. eylesi
- Anopheles (Anopheles) sacharovi - P. vivax
- Anopheles (Cellia) sergentii - P. vivax
- Anopheles sinensis Anopheles (Anopheles) sinensis - P. eylesi, P. fieldi, P. vivax
- Anopheles (Cellia) stephensi - P. cynomogli, P. fieldi, P. inui, P. vivax
- Anopheles (Cellia) sundaicus - P. eylesi, P. vivax, P. youngei
- Anopheles (Cellia) superpictus - P. vivax
- Anopheles (Cellia) tessellatus - P. falciparum, P. vivax
- Anopheles (Nyssorhynchus) triannulatus - P. falciparum, P. vivax
- Anopheles (Nyssorhynchus) trinkae - P. vivax
- Anopheles (Anopheles) umbrosus - P. eylesi
- Anopheles (Cellia) vagus - P. eylesi, P. fieldi
References[edit]
- ^ Coatney GR, Chin W, Contacos PG, King HK; Chin; Contacos; King (1966). "Plasmodium inui, a quartan-type malaria parasite of Old World monkeys transmissible to man". J Parasitol 52 (4): 660–666. doi:10.2307/3276423. JSTOR 3276423.
- ^ Contacos PG, Coatney GR, Orihel TC, Collins WE, Chin W; Coatney; Orihel; Collins; Chin; Jeter (1970). "Transmission of Plasmodium schwetzi from the chimpanzee to man by mosquito bite". Am J Trop Med Hyg 19 (2): 190–5. PMID 5443069.
- ^ Rodhain J, Dellaert R (1955). "Contribution a l'etude de Plasmodium schwetzi E. Brumpt (2eme note). Transmission de Plasmodium schwetzi a l'homme". Ann. Soc. Belg. Med. Trop. 35: 757–75.
- ^ Tsukamoto M (1977). "An imported human malarial case characterized by severe multiple infections of the red blood cells". Ann. Trop. Med. Parasit. 19 (2): 95–104.
- ^ Russel PF (1928). "Plasmodium tenue (Stephens): A review of the literature and a case report". Am. J. Trop. Med. s1–8 (5): 449–79.
- ^ a b c Prugnolle F, Durand P, Neel C, Ollomo B, Ayala FJ, Arnathau C, Etienne L, Mpoudi-Ngole E, Nkoghe D et al. (2010). "African great apes are natural hosts of multiple related malaria species, including Plasmodium falciparum". Proc. Natl. Acad. Sci. USA 107 (4): 1458–1463. doi:10.1073/pnas.0914440107. PMC 2824423. PMID 20133889.
- ^ a b Krief S, Escalante AA, Pacheco MA, Mugisha L, André C, Halbwax M, Fischer A, Krief JM, Kasenene JM, Crandfield M, Cornejo OE, Chavatte JM, Lin C, Letourneur F, Grüner AC, McCutchan TF, Rénia L, Snounou G (2010) On the Diversity of malaria parasites in African apes and the origin of Plasmodium falciparum from bonobos. PloS Pathog. 12;6(2):e1000765
- ^ a b c Duarte AM, Malafronte Rdos S, Cerutti C; Malafronte Rdos; Cerutti Jr; Curado; De Paiva; Maeda; Yamasaki; Summa; Neves Ddo; De Oliveira; Gomes Ade (August 2008). "Natural Plasmodium infections in Brazilian wild monkeys: Reservoirs for human infections?". Acta Trop. 107 (2): 179–85. doi:10.1016/j.actatropica.2008.05.020. PMID 18620330.
- ^ Ayouba A, Mouacha F, Learn GH, Mpoudi-Ngole E, Rayner JC, Sharp PM, Hahn BH, Delaporte E, Peeters M (2012) Ubiquitous Hepatocystis infections, but no evidence of Plasmodium falciparum-like malarial parasites in wild greater spot-nosed monkeys (Cercopithecus nictitans). Int J Parasitol
- ^ Ollomo B, Durand P, Prugnolle F; Durand; Prugnolle; Douzery; Arnathau; Nkoghe; Leroy; Renaud (May 2009). "A new malaria agent in African hominids". PLoS Pathog. 5 (5): e1000446. doi:10.1371/journal.ppat.1000446. PMC 2680981. PMID 19478877.
- ^ a b c d e f g h i Kaiser M, Lowa A, Ulrich M, Ellerbrok H, Goffe AS, Blasse A, Zommers Z, Couacy-Hymann E, Babweteera F et al. (Dec 2010). "Wild chimpanzees infected with 5 Plasmodium species". Emerg Infect Dis 16 (12): 1956–1959. doi:10.3201/eid1612.100424. PMID 21122230.
- ^ Da Silva, Araújo M; Messias, MR; Figueiró, MR; Gil, LH; Probst, CM; de Medeiros Vidal, N; Katsuragawa, TH; Krieger, MA et al. (2013). "Natural Plasmodium infection in monkeys in the state of Rondonia (Brazilian Western Amazon)". Malar J 12 (1): 180. doi:10.1186/1475-2875-12-180.
- ^ Rodhain J (1940) Les plasmodiums des anthropoids de l'Afrique centrale et leurs relations avec les plasmodiums humains. Récepticité de l'homme au Plasmodium malariae. (Plasmodium rodhaini Brumpt) du chimpanzé. C. R. Soc. Biol. 133:276-277
- ^ Rodhain J and Dellaert R (1943) L'infection á Plasmodium malariae du chimpanzé chez l'homme. Etude d'une premiére souche isolée de l'anthropoide Pan satyrus verus. Ann. Soc. Belge. Med. Trop. 23:19-46
- ^ Hayakawa T, Arisue N, Udono T; Arisue; Udono; Hirai; Sattabongkot; Toyama; Tsuboi; Horii; Tanabe (2009). "Identification of Plasmodium malariae, a human malaria parasite, in imported chimpanzees". PLoS ONE 4 (10): e7412. doi:10.1371/journal.pone.0007412. PMC 2756624. PMID 19823579.
- ^ Collins WE, Contacos PG; Contacos (1969). "Infectivity of Plasmodium malariae in the Aotus trivirgatus monkey to Anopheles freeborni mosquitoes". J Parasitol 55 (6): 1253–1257. doi:10.2307/3277270. JSTOR 3277270. PMID 4982903.
- ^ Pacheco MA, Cranfield M, Cameron K, Escalante AA (2013) Malarial parasite diversity in chimpanzees: the value of comparative approaches to ascertain the evolution of Plasmodium falciparum antigens. Malar J 12(1):328
- ^ Reid MJ, Ursic R, Cooper D; Ursic; Cooper; Nazzari; Griffiths; Galdikas; Garriga; Skinner; Lowenberger (December 2006). "Transmission of human and macaque Plasmodium spp. to ex-captive orangutans in Kalimantan, Indonesia". Emerging Infect. Dis. 12 (12): 1902–8. doi:10.3201/eid1212.060191. PMC 3291341. PMID 17326942.
- ^ Prugnolle F, Rougeron V, Becquart P, Berry A, Makanga B, Rahola N, Arnathau C, Ngoubangoye B, Menard S, Willaume E, Ayala FJ, Fontenille D, Ollomo B, Durand P, Paupy C, Renaud F (2013) Diversity, host switching and evolution of Plasmodium vivax infecting African great apes. Proc Natl Acad Sci USA
- ^ Duval L, Nerrienet E, Rousset D; Nerrienet; Rousset; Sadeuh Mba; Houze; Fourment; Le Bras; Robert; Ariey (2009). "Chimpanzee malaria parasites related to Plasmodium ovale in Africa". PLoS ONE 4 (5): e5520. doi:10.1371/journal.pone.0005520. PMC 2677663. PMID 19436742.

- ^ Sutherland CJ, Tanomsing N, Nolder D, Oguike M, Jennison C, Pukrittayakamee S, Dolecek C, Hien TT, do Rosário VE, Arez AP, Pinto J, Michon P, Escalante AA, Nosten F, Burke M, Lee R, Blaze M, Otto TD, Barnwell JW, Pain A, Williams J, White NJ, Day NP, Snounou G, Lockhart PJ, Chiodini PL, Imwong M, Polley SD; Tanomsing; Nolder; Oguike; Jennison; Pukrittayakamee; Dolecek; Hien; Do Rosário; Arez; Pinto; Michon; Escalante; Nosten; Burke; Lee; Blaze; Otto; Barnwell; Pain; Williams; White; Day; Snounou; Lockhart; Chiodini; Imwong; Polley (2010). "Two nonrecombining sympatric forms of the human malaria parasite Plasmodium ovale occur globally". J Infect Dis 201 (10): 1544–50. doi:10.1086/652240. PMID 20380562.
- ^ Coatney GR, Roudabush RL; Roudabush (1936). "A catalog and host-index of the genus Plasmodium". J. Parasitol. 22 (4): 338–53. doi:10.2307/3271859. JSTOR 3271859.
- ^ Collins WE, Sullivan JS, Nace D, Williams T, Williams A, Barnwell JW; Sullivan; Nace; Williams; Williams; Barnwell (February 2008). "Observations on the sporozoite transmission of Plasmodium vivax to monkeys". J. Parasitol. 94 (1): 287–8. doi:10.1645/GE-1283.1. PMID 18372652.
- ^ Collins WE, Richardson BB, Morris CL, Sullivan JS, Galland GG; Richardson; Morris; Sullivan; Galland (July 1998). "Salvador II strain of Plasmodium vivax in Aotus monkeys and mosquitoes for transmission-blocking vaccine trials". Am. J. Trop. Med. Hyg. 59 (1): 29–34. PMID 9684622.
- ^ Collins WE, Sullivan JS, Nace D; Sullivan; Nace; Williams; Sullivan; Galland; Grady; Bounngaseng (April 2002). "Experimental infection of Anopheles farauti with different species of Plasmodium". J. Parasitol. 88 (2): 295–8. doi:10.1645/0022-3395(2002)088[0295:EIOAFW]2.0.CO;2. JSTOR 3285576. PMID 12054000.
- ^ Collins WE, Morris CL, Richardson BB, Sullivan JS, Galland GG; Morris; Richardson; Sullivan; Galland (August 1994). "Further studies on the sporozoite transmission of the Salvador I strain of Plasmodium vivax". J. Parasitol. 80 (4): 512–7. doi:10.2307/3283184. JSTOR 3283184. PMID 8064516.
- ^ Wharton RH, Eyles DE.; Eyles (1961). "Anopheles hackeri, a vector of Plasmodium knowlesi in Malaya". Science 134 (3474): 279–80. doi:10.1126/science.134.3474.279. PMID 13784726.
- ^ Vythilingam I, Tan CH, Asmad M, Chan ST, Lee KS, Singh B.; Tan; Asmad; Chan; Lee; Singh (2006). "Natural transmission of Plasmodium knowlesi to humans by Anopheles latens in Sarawak, Malaysia". Trans R Soc Trop Med Hyg 100 (11): 1087–88. doi:10.1016/j.trstmh.2006.02.006. PMID 16725166.
- ^ Tan CH, Vythilingam I, Matusop A, Chan ST, Singh B; Vythilingam; Matusop; Chan; Singh (2008). "Bionomics of Anopheles latens in Kapit, Sarawak, Malaysian Borneo in relation to the transmission of zoonotic simian malaria parasite Plasmodium knowlesi". Malar. J. 7: 52. doi:10.1186/1475-2875-7-52. PMC 2292735. PMID 18377652.
Paraplasmodium is a subgenus of the genus Plasmodium - all of which are parastic protozoa. The subgenus was created by Telford in 1988. Species in this subgenus infect lizards.
Diagnostic features
Species in the subgenus Paraplasmodium have the following characteristics:
The gametocytes are large.
The schizonts are medium size.
Exoerythrocytic schizonts may be produced in both fixed and wandering host cells.
Note:
One species (Plasmodium mexicanum) in this genus can undergo normal sporogony in a psychodid fly (Lutzomyia vexatrix).
Species in this subgenus
References
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| This medical article is a stub. You can help Wikipedia by expanding it. |
| This infectious disease article is a stub. You can help Wikipedia by expanding it. |
| This chromalveolate article is a stub. You can help Wikipedia by expanding it. |
| License | http://creativecommons.org/licenses/by-sa/3.0/ |
| Rights holder/Author | Wikipedia |
| Source | http://en.wikipedia.org/w/index.php?title=Paraplasmodium&oldid=539993119 |